



Next: Accidental background
Up: Systematic issues
Previous: In-situ monitoring of diffusion
Contents
For C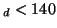 ppm
ppm 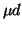 diffusion is completely determined by
diffusion is completely determined by 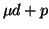 scattering and
scattering and 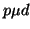 formation, as
formation, as 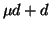 scattering is too infrequent.
Thus the shape of the distribution and of p(r,t) is independent of C
scattering is too infrequent.
Thus the shape of the distribution and of p(r,t) is independent of C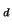 .
As seen from table 9, in the limit of small radial cuts the correction is simply
.
As seen from table 9, in the limit of small radial cuts the correction is simply
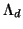 , i.e. proportional to C
, i.e. proportional to C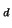 . Thus measuring of the
decay rate
. Thus measuring of the
decay rate 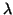 at two different deuterium concentrations C
at two different deuterium concentrations C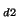 /C
/C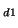 =C
allows an extrapolation to C
=C
allows an extrapolation to C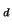 =0. The error of the extrapolated value
has the following three sources of uncertainties:
=0. The error of the extrapolated value
has the following three sources of uncertainties:
The first and second come directly from 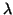 measurement errors
at different deuterium concentrations
measurement errors
at different deuterium concentrations 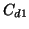 and
and 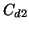 , respectively.
The third one comes from the error of determination of the ratio C. The total
error of extrapolated value of
, respectively.
The third one comes from the error of determination of the ratio C. The total
error of extrapolated value of
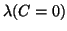 is
is
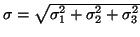 .
To decrease the error of extrapolation we need a measurement for large
.
To decrease the error of extrapolation we need a measurement for large
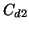 (up to
(up to 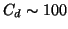 ppm, where the correction is still
linear to
ppm, where the correction is still
linear to 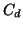 ). It is important that at
). It is important that at 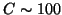 the measurement
at the second deuterium concentration could be done with only
the measurement
at the second deuterium concentration could be done with only 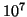 events without loss of precision of the extrapolated value. Then the resulting
precision will depend only on the first and the third contributions. See table 10
for
events without loss of precision of the extrapolated value. Then the resulting
precision will depend only on the first and the third contributions. See table 10
for 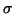 as the function of
as the function of 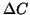 for two
for two 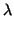 measurements, for
measurements, for 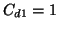 ppm and 100 ppm.
ppm and 100 ppm.
In conclusion, for tight tracking we need an additional measurement of 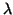 at deuterium concentration
at deuterium concentration 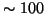 ppm and a precison of better than
ppm and a precison of better than 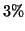 for ratio of concentrations
for ratio of concentrations
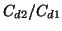 . If we enlarge the
tracking radius to 5 cm we only need about 6% precison for this ratio.
. If we enlarge the
tracking radius to 5 cm we only need about 6% precison for this ratio.




Next: Accidental background
Up: Systematic issues
Previous: In-situ monitoring of diffusion
Contents
Peter Kammel
2001-02-04
![]() ppm
ppm ![]() diffusion is completely determined by
diffusion is completely determined by ![]() scattering and
scattering and ![]() formation, as
formation, as ![]() scattering is too infrequent.
Thus the shape of the distribution and of p(r,t) is independent of C
scattering is too infrequent.
Thus the shape of the distribution and of p(r,t) is independent of C![]() .
As seen from table 9, in the limit of small radial cuts the correction is simply
.
As seen from table 9, in the limit of small radial cuts the correction is simply
![]() , i.e. proportional to C
, i.e. proportional to C![]() . Thus measuring of the
decay rate
. Thus measuring of the
decay rate ![]() at two different deuterium concentrations C
at two different deuterium concentrations C![]() /C
/C![]() =C
allows an extrapolation to C
=C
allows an extrapolation to C![]() =0. The error of the extrapolated value
has the following three sources of uncertainties:
=0. The error of the extrapolated value
has the following three sources of uncertainties:
![]() at deuterium concentration
at deuterium concentration ![]() ppm and a precison of better than
ppm and a precison of better than ![]() for ratio of concentrations
for ratio of concentrations
![]() . If we enlarge the
tracking radius to 5 cm we only need about 6% precison for this ratio.
. If we enlarge the
tracking radius to 5 cm we only need about 6% precison for this ratio.